Neutrog’s Microbiologist and R&D Manager, Dr. Uwe Stroeher’s recent article called ‘Nitrogen Fixation in Agriculture by Mutualistic and Free Living Bacteria’ was published in the autumn issue of the Australian Agronomist magazine.
Uwe discusses the role nitrogen fixation plays within plant tissues of legumes or within the soil itself by free living bacteria, and how bacteria can turn atmospheric nitrogen into a plant-useable form such as ammonia.
Nitrogen, together with potassium and phosphate, are the most widely used fertilisers in the world. Until the advent of the Haber-Bosch process in the early 1900s which allows the chemical synthesis of nitrogen fertilisers, the world was reliant on organic fertilisers and the ability of bacteria to fix atmospheric nitrogen (Erisman et al., 2008).
There is no doubt that without the use of chemical nitrogen we would not be able to feed the world’s population, however nitrogen from chemical fertilisers comes at enormous cost, both financially and environmentally. Production of nitrogen fertilisers alone requires over 1% of the worlds’ energy, and it’s this high energy requirement that is at the crux of turning atmospheric nitrogen into a plant-useable form such as ammonia.
In the atmosphere, nitrogen is a molecule of two tightly bound nitrogen atoms which need to be split to combine with hydrogen to form ammonia or oxygen – which then forms nitrites or nitrates. This occurs in nature during lightning, where the molecules are torn apart and can then combine with oxygen. Alternatively, it can occur via a biological process driven by a number of bacterial species. In the soil, nitrogen fixation occurs either within plant tissues of legumes, or within the soil itself by free living bacteria.
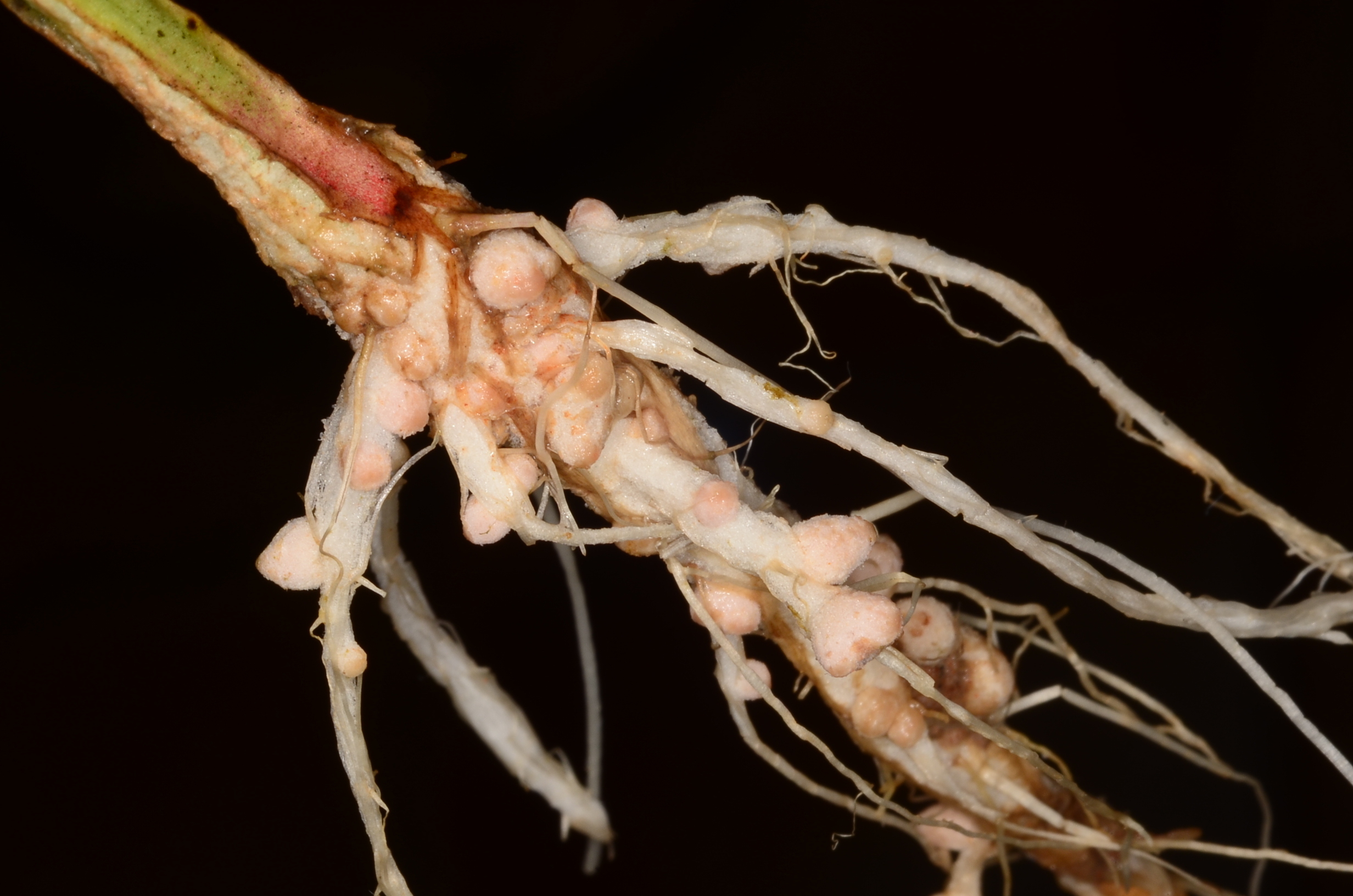
Most people are aware of the nodules that form on the roots of legumes – it is within these structures that bacteria fix nitrogen
(Fig. 1). For each legume species there exists a distinct set of Rhizobium species which associates with the roots and forms these nodules. For example, bacteria found in peas will not associate with beans or lentils. This interaction is extremely specific, and is driven by the bacteria and the plant, and only occurs when the plant is under limited nitrogen conditions (Maróti and Kondorosi 2014).
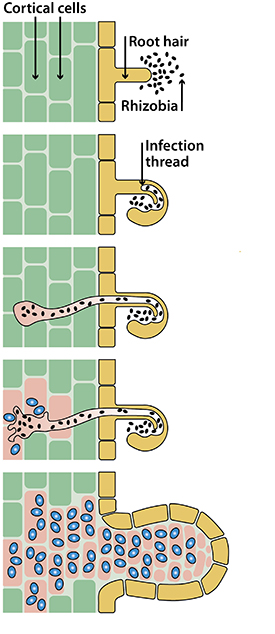
Initially, the plant roots release compounds known as flavonoids, which attract soil bacteria such as Rhizobia that are capable of fixing nitrogen, and the bacteria start to multiply near the root (Fig. 2). This process is not 100% specific, but can also recruit other bacteria which may be undesirable. Therefore, the plant ‘looks’ for a bacterial signal, which communicates to the plant that the bacteria is capable of nodule formation and nitrogen fixation. This signal is called the nodulation factor, and causes the root hair to curl, forming an infection thread.
A mutualistic relationship is then established between the plant and bacteria, and the bacteria enters the root cortex via the infection thread. Once the bacteria are released from the infection thread, they become bacteroids and multiply (refer to the blue shapes in Fig. 2). Similarly, the plant root cells surrounding the bacteroids start to divide and begin nodule formation.
Although nodules may be present, the process of nitrogen fixation requires some additional factors to be met. Firstly, the plant must be in a low nitrogen environment. If levels of nitrogen are high, nodulation may not occur, or the nodules will not fix nitrogen. Secondly, nitrogen fixation only occurs if it is within the plant nodules or in the soil where there is an environment of low oxygen. To achieve this within the nodules, the plant produces a protein known as leghaemoglobin (also known as legoglobin), which has a very high affinity for oxygen, and provides a low-oxygen environment for nitrogen fixation. The requirement for low oxygen is due to the sensitivity of the nitrogenase enzymes, which are essential for nitrogen fixation and provided by the bacteria.
Similar to human haemoglobin, Leghaemoglobin is red in colour, and only nodules that show a pinkish or red/brown internal colouration are capable of fixing nitrogen. Nitrogen fixation is tightly controlled and regulated because it is extremely energy intensive, and only occurs when required. Therefore, whereas the plant obtains a ready source of nitrogen, the bacteria is given carbohydrates to produce the energy for nitrogen fixation and for general bacterial metabolism, and the nodule provides a niche that is free of competing microbes, giving the Rhizobium a distinct advantage.
Legumes are generally grown as either a high-protein livestock food or a crop, and can fix in excess of 200kg of nitrogen per hectare, however not all of this nitrogen is available for subsequent crops, as the plant depletes the ammonia levels within the nodule during flower and seed production.
If the legumes are grown for their ability to fix nitrogen as a cover crop and to enrich the soil for a subsequent harvest, then this crop should be ploughed back into the ground just after the first sign of flower setting. After this point, nitrogen in the nodules is starting to be drawn out for flowering and seeds. For some legumes, 40% of the fixed nitrogen is eventually trapped in the seed. Once ploughed back into the soil, the plant, roots and nodules break down, thereby releasing nitrogen for other crops.
Surprisingly, nitrogen fixation is not limited to the mutualistic relationship discussed above, but is also undertaken by a number of free living soil bacteria. Amongst the most important are species of Azotobacter and Clostridium – their ability to fix nitrogen is lower (around 30kg per hectare), yet they still represent significant nitrogen fixation. The requirements to fix nitrogen by these bacteria is no different to the Rhizobium. Within the soil – and in particular the rhizosphere – there are plant-secreted carbohydrates which are used in part to energise nitrogen fixation. The issue of low oxygen concentration is overcome in Azotobacter by the production of a slime layer which lowers the intracellular oxygen concentration. As Clostridium grow in anaerobic environments, oxygen is not an issue. In essence, nitrogen fixation occurs in almost all microbiologically-active soils and is not restricted to legumes.
The last group of nitrogen-fixing bacteria are the cyanobacteria – these play a significant role in nitrogen fixation in wet soils or in environments such as rice paddies. In this instance, they fix approximately 20-30 kg of nitrogen per hectare. Cyanobacteria are photosynthetic so they are not dependent on an external source of carbohydrates for their energy. Furthermore, they have specialised structures called heterocycsts where low levels of oxygen are produced and nitrogen fixation occurs.
Thus, in order to maximise nitrogen fixation, there are a number of factors to consider. For legumes, various Rhizobium inoculants are available to increase nodule formation, however this will do little unless other factors are met. The most obvious is to ensure nitrogen fertilisers are used at a level which requires legumes to fix atmospheric nitrogen. This is also the case for free living nitrogen-fixing bacteria. Secondly, this is a biological reaction, so an environment that encourages microbial diversity is best. Furthermore, if legumes are stressed due to environmental factors or an imbalance of nutrients, then nitrogen fixation suffers. Ensuring a ready supply of phosphorus, potassium, sulphur, zinc, iron, cobalt and molybdenum will enhance nitrogen fixation. An organic fertiliser such as Neutrog’s Rapid Raiser will provide these nutrient requirements without adding excessive nitrogen.
References:
Erisman, J. W., Sutton, M. A., Galloway, J., Klimont, Z., and Winiwarter, W. (2008). How a century of ammonia synthesis changed the world. Nature Geoscience, 1, 636-639.
Maróti, G., and Kondorosi, E. (2014) Nitrogen-fixing Rhizobium-legume symbiosis: are polyploidy and host-peptide governed symbiont differentiation general principles of endosymbiosis? Frontiers of Microbiology, 5, 236
